First of all, Modum is doing great, we managed to stay healthy and fully operational. We have not only managed to deliver on all our customer projects over the past months, we have also adapted our organization in the face of COVID-19 implications to our marketing and sales activities. Thanks to the thorough marketing work we did last year in refining our way of promoting Modum and its solutions directly and through numerous partners, we were able to mitigate a large part of the impact by now and can continue to plan for us and our customers as long-term as we are used to do.
Modum’s market approach
We are continuing to establish Modum and our solution as a capable and competitive player in the pharma supply chain industry. We have built active relationships with leaders of about 120 pharma brand owners and about 50 pharma wholesalers. We have learned that our approach of not just supplying measurement devices but to offer holistic supply chain visibility and steering systems that are well-integrated into the leading IT systems of pharma brand owners and distributors, is well-appreciated. In addition, supply chain and quality professionals see the value of not just measuring supply chains with MODsense but also to fully leverage the data collected with our AI-driven MODsight analytics software and to feed these insights in a trusted way either to other supply chain stakeholders or back in to their own logistics processes – using integration and data management options we label under the term MODlink.
So, where do we stand with regards to applications and market traction with respect to each solution area?
MODsense
Today, we are pursuing two main applications of MODsense – monitoring of (intercontinental) warehouse to warehouse shipments, and monitoring of national distribution of sensitive goods all the way to the point of sale or dispense (e.g. hospitals or pharmacies), as launched last year with Swiss Post.
In the last year we have not just continued to evolve our solution in collaboration with Swiss Post and their customers, but we have also established scalable use cases for the same core system with well-known pharma manufacturers and distributors in Europe and Asia. With them, we are now in the process of industrializing and rolling out the system, mostly for pharmaceutical products and drug kits for clinical trials but also for samples of medical laboratories.
The results we can demonstrate there, show that MODsense is clearly outperforming legacy solutions, being at the sweet spot of cost-efficiency but still very effective and simple to use.
Hence, we are building out our MODsense ecosystem on in all dimensions. On the softwareside, where we recently released improved mobile and desktop apps for even smoother workflows that allow users to add shipment-specific meta-data, improved regulatory compliance with advanced audit trails and enabled Modum-managed logger inventory features. On the hardware side, we now also offer mobile gateways that completely automate the interaction with our data loggers, further reducing the already low handling cost to a point where MODsense becomes a uniquely attractive solution for data collection at point of dispense or at designated cross-docking locations.
So, despite our desire to speed things up in general, we are happy how MODsense is evolving for the secondary distribution use case!
On top of this, we expanded MODsense to enable real-time tracking and monitoring of international and intercontinental shipments in the primary distribution network of pharma brand owners. For this purpose, we are expanding our MODsense solution portfolio with the new XL-RT data logger which is not only capable of measuring temperature, but also other conditions such as humidity, ambient light and more. In addition, MODsense XL-RT is also able to determine its location and to continuously communicate all of the above data via cellular communication networks on a global scale.

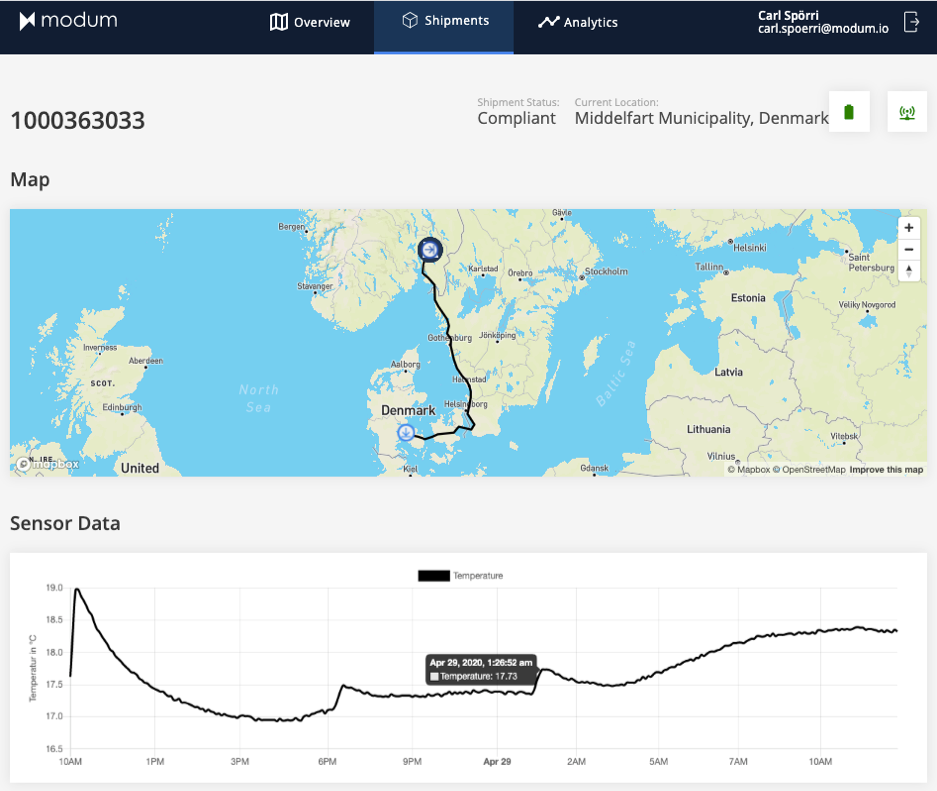
In summary, with our Gateways and the XL-RT data logger, we are expanding MODsense into the industry’s most powerful and modular end-to-end monitoring system. With the full ownership of both hardware and software, we are unlocking a huge potential in designing truly process-centric supply chain monitoring systems. And this does not stop at monitoring the transportation of pharmaceutical goods but expands to permanent temperature and humidity monitoring of warehouses or tracking of high-value equipment not necessarily related to pharmaceutical applications.
Read more about MODsight and MODlink here!
I hope this provides some good insight into where Modum stands and what we are up to in the coming years and the customer and prospect portfolio we built up so far, gives us the confidence that we are doing the right thing.
Today, we are thankful that we see business interactions coming out of lockdown again and I believe in the long run, COVID-19 sped up supply chain digitalization and unearthed challenges to solve.
We are ready and eager to help solve them.
Stay healthy!
Simon Dössegger, CEO Modum